AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Atomic theory of matter4/17/2024  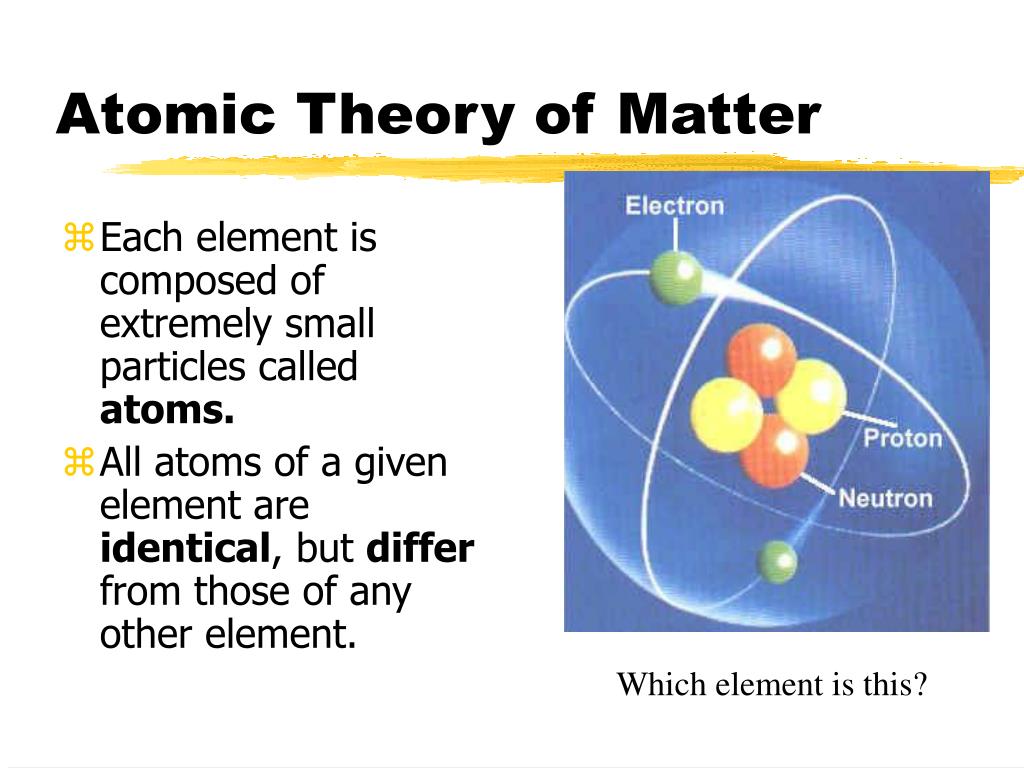 
The law of conservation of mass states that the total mass present before a chemical reaction is the same as the total mass present after the chemical reaction. Explore the postulates of Daltons theory, the laws he based it on, and the modifications made since then. The third part says compounds are combinations of two or more different types of atoms. Learn about the first complete attempt to describe all matter in terms of atoms and their properties. Previously, we defined an atom as the smallest part of an element that maintains the identity of that element. 1 ), is a fundamental concept that states that all elements are composed of atoms. The second part of the theory says all atoms of a given element are identical in mass and properties. The modern atomic theory, proposed about 1803 by the English chemist John Dalton (Figure 2.2.1 2.2. The concept that matter is composed of discrete particles is an ancient idea, but gained scientific credence in the 18th and 19th centuries when scientists found it could explain the behaviors of gases and how chemical elements reacted with each other. This unit explores the atomic theory of matter, the foundational premise of chemistry. Six postulates are involved in Daltons Atomic Theory: All matter consists of indivisible particles called atoms. Dalton thought that atoms were the smallest units of matter tiny, hard spheres that could not be broken down any further. However, one of his underlying assumptions was later shown to be incorrect. 
Scientists did not account for the gases that play a critical role in this reaction. The first part of his theory states that all matter is made of atoms, which are indivisible. Atomic theory is the scientific theory that matter is composed of particles called atoms. Daltons ideas proved foundational to modern atomic theory. \) shows that the burning of word does follow the law of conservation of mass. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
